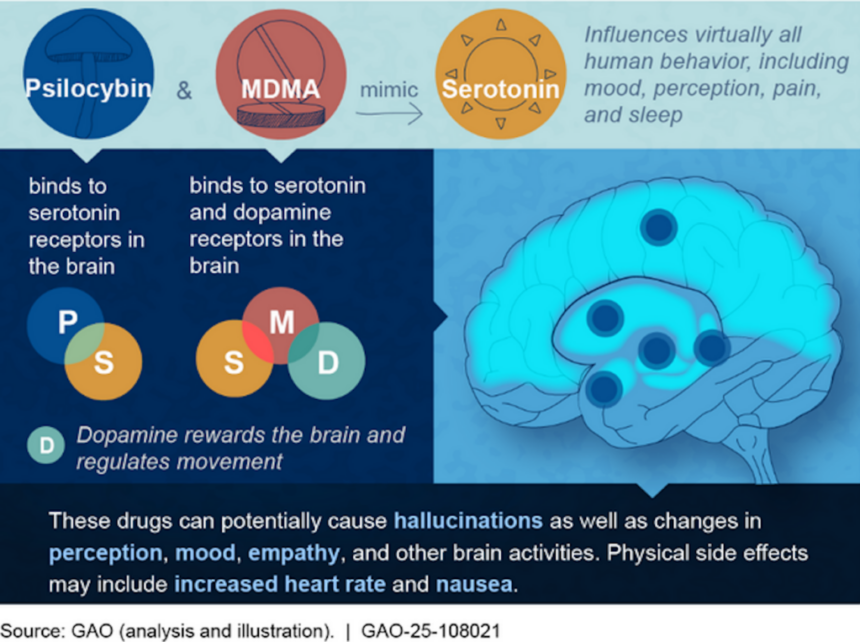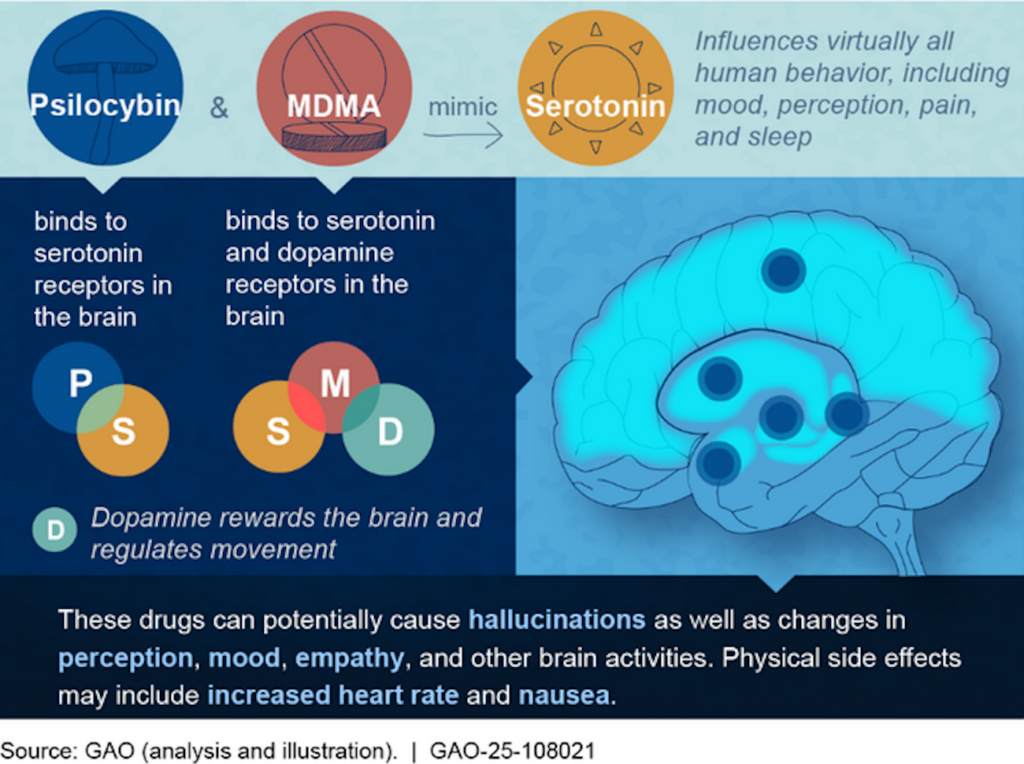
A federal agency has published a short report on the medical use of psychedelics, finding that their ability to “change a person’s perceptions and sense of self” can make for “promising medical applications.”
The paper, from the Government Accountability Office (GAO), says there’s a need for further research, but it also notes that because psychedelics such as LSD, MDMA and psilocybin remain Schedule I controlled substances, securing permission to carry out that research can be a challenge.
“To conduct research on these drugs, scientists need to follow several steps,” the report from GAO, which is often referred to as Congress’s “watchdog,” states in a section about ongoing challenges. “These include obtaining permission from the U.S. Drug Enforcement Administration, finding clinical grade drugs to test, and identifying appropriate spaces in which to test and store these drugs.”
“Difficulties associated with conducting large, blind trials of psychedelics have limited researchers’ ability to determine the safety and effectiveness of these drugs,” GAO continues, “which is required for them to gain approval from the Food and Drug Administration (FDA). FDA approval is generally required before prescription drugs can be marketed for sale in the U.S.”
Despite the obstacles, the paper acknowledges that hundreds of
Read full article on Marijuana Moment